Biological Sciences
The Maternal-Fetal Immune Dialogue: Systemic and Local Mechanisms

Aditi Singh '28
Mar 9, 2026
Overview
During pregnancy, interactions between the fetus and mother are bidirectional. The fetus presents antigens that reflect paternal characteristics, which may trigger a damaging maternal immune response. Thus, the mother must modulate her immune and metabolic systems to accommodate the growth of the fetus (Thellin et al., 2003).
The placenta is the site of the materno-fetal graft and has a particularly unique immune system. The placental immune population has different surface markers than other immune cells at the same time of gestation. Also unique to the placenta are decidual macrophages, which function as an IL10 and IL-1R antagonists. Additionally, the production of glycoprotein OX-2 within the placenta inhibits macrophages and decreases fgl2, a compound that triggers abortion through placental thrombosis (Thellin et al., 2003).
The other relevant anatomical site is the endometrium, which transforms into the decidua during pregnancy. This process of decidualization is mediated by progesterone and results in marked morphological and functional differentiation that is essential for a successful pregnancy (Hidetaka, 2018). Decidualization also results in substantial immune changes.
Maintaining the delicate balance of the immune system requires simultaneously ensuring fetal viability and maternal protection; this adaptation is mediated by both local and systemic mechanisms. Initial hypotheses surrounding maternal tolerance suggest that pregnancy is primarily a Th2 phenomenon, which are cells that produce interleukins 4, 5, 6, 9, and 10, reducing the production of pro-inflammatory molecules that endanger fetal survival (Warning et al., 2011). Essentially, maternal Th1 cells are inhibited via NF-κB during pregnancy, while Th2 cells that mediate antibody responses are increased to prevent complete immunosuppression (Thellin et al., 2003).
However, new evidence suggests that success in pregnancy and maintenance of the materno-fetal graft also require involvement of cytokines typically associated with Th1 responses (Warning et al., 2011. Considering that the unique immune environment surrounding the maternal-fetal relationship comprises multiple components and is indicative of pregnancy outcomes, it is important to examine the complexities of this relationship on both systemic and local levels.
Systemic Response
The maternal immune system is modulated systemically through progesterone and human leukocyte antigens (HLAs). Progesterone, produced in large quantities by the placenta, is able to lower immune responses in the maternal environment by limiting the activation of murine dendritic cells, macrophages, and natural killer (NK) cells (Raghupathy et. al, 2022). The progesterone-induced blocking factor (PIBF) is a downstream mediator of progesterone responsible for many of its immunological effects. A mouse model demonstrated that PIBF deficiency is associated with increased decidual and peripheral NK activity, as well as the differentiation of more T cells into Th1 cells (Csabai 2020). Notably, there are two receptors involved in progesterone-based modulation: PR-A, which plays a role in implantation and decidualization, and PR-B, which is needed for the development of the mammary gland (Raghupathy et. al, 2022). Moreover, fetal antigens or cells pass into the maternal blood circulation and lymph organs, acting as tolerogens and inducing peripheral tolerance.
Syncytiotrophoblasts, fetal cells that are in direct contact with the maternal blood supply, do not express HLA class I or II molecules (Rao et al., 2022). Considering that HLA class I and II molecules are responsible for presenting antigens to T lymphocytes, this creates an immune-privileged physical barrier. Together, these mechanisms create a maternal environment in which immune cells are unable to function due to antigens acting as tolerogens and the inability of T cells to recognize potential pathogens.
Localized immune responses to pregnancy are more robustly understood.
Local Responses: HLA
Fetal-derived factors like HLA-C, HLA-G, and Treg cells are expressed at the maternal-fetal interface. The trophoblast is a thin layer of cells that protects the developing embryo and helps it attach to the wall of the uterus. As a result, the immune expression at the trophoblast is a crucial factor in preventing spontaneous abortion; if the cells at this interface begin to cause inflammation around the developing embryo, this can result in high-risk situations. As an HLA Class I molecule expressed on the trophoblast, HLA-C is recognized by maternal NK and T cells, preventing harmful immune responses. Interestingly, pregnancies in which the mother and fetus have similar HLA-C profiles have an increased risk of spontaneous abortion, suggesting that some level of immunological difference between the mother and fetus is necessary to invoke other crucial immune responses (Warning et. al, 2011).
HLA-G cells, responsible for modulating T cells by downregulating CD4 and CD8 expression, are also found on the trophoblast. Through this downregulation, HLA-G increases the proportion of HLA-DRC+ T cells and generates suppressor T cells (Treg cells) by producing TBG-β (Rao et. al, 2022). Studies show that an increase in plasmatic HLA-G cells is associated with increased materno-fetal graft survival. Treg cells generate a positive feedback loop, upregulating HLA-G expression by the trophoblast. HLA G cells then bind to CD8 T cells (non-suppressor, immunoactive T cells), leading to inactivation and further T cell apoptosis (Warning et al., 2011).
Local Responses: NK Cells
Natural killer (NK) cells are cytotoxic and are instrumental in clearing virus-infected cells (Warning et al., 2011). Inhibitory and activating. There are three broad categories of NK cells: peripheral NK (pNK), endometrial NK (eNK), and decidual NK (dNK).
pNKs are CD56dim/CD16+, meaning that they have poor expression of the CD56 marker and high presence of CD16 (Warning et. al, 2011. CD16 is involved in the lysis of target cells; specifically, it binds to IgG antibodies attached to target cells. When the CD16 engages these antibodies, it generates antibody-dependent cell-mediated cytotoxicity (ADCC), enabling pNKs to kill the target cells.
dNKs and eNKs are part of a broader group of uterine NKs (uNKs). uNKs collaborate to remodel maternal spiral arteries into wide vessels that support blood flow to the placenta. This process operates through cytokines and the production of factors like VEGF and L-8 (Warning et. al, 2011). Beyond its role in fetal nutrition, evidence suggests that the specifics of this blood flow are more important than once thought; in fact, the force of blood moving against vessel walls provides signaling for fetal development (Morley 2021). This means that the precise modulation of uNKs is incredibly important.
eNKs, present prior to pregnancy, are vital in preparing the endometrium for fetal implantation. Unlike pNKs, eNKs are CD56dim/CD16-, so they display reduced cytotoxicity. Their numbers rise during the secretory phase of the menstrual cycle, and upon implantation IL-15 enables their differentiation into decidual NKs (Lee 2011).
dNKs are CD56bright/CD16-, so they have reduced cytotoxicity and primarily function to produce cytokines. The majority of the dNKs are located at the decidua basalis– the site of trophoblast invasion (Warning et. al, 2011. The growth factors and chemokines within the decidua promote vascular remodeling, a process that helps accommodate increased blood flow to the placenta. Conversely, the inhibition of dNK cells may increase the likelihood of preeclampsia (Wei 2023). While NK cells are generally considered cytotoxic, these cells at the maternal-fetal interface are recruited by the trophoblast to perform important functions in the decidua.
Because of this functionality, it is documented that NK levels increase throughout pregnancy, and this change is contingent on progesterone. NK cells make up 70% of mononuclear cells in the pregnant womb, and their numbers decrease from mid-gestation to term (Sharma 2015).
It is also known that NKs have two types of polarization–-NK1 and NK2– and both of these polarization types are present in eNK, dNK, and pNK cells. NK1 cells engage in pro-inflammatory action, while NK2 cells produce interleukins (ILs) with anti-inflammatory action (Rao et. al, 2022). It is intuitive, then, that dNKs have a strong profile of NK2 cells. While eNK populations are slightly NK1-leaning, during healthy pregnancies, this profile shifts towards being primarily NK2 (Rao et. al, 2022).
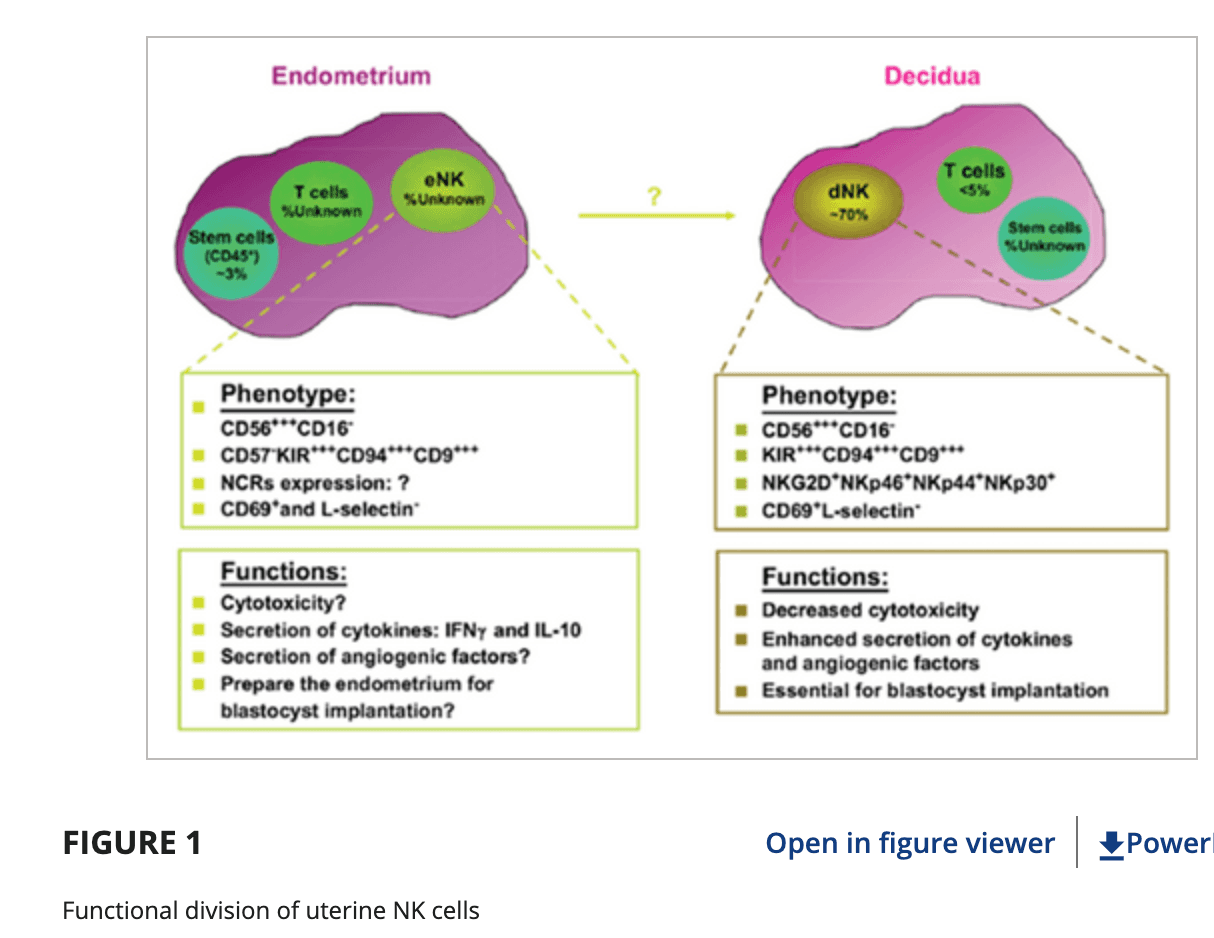
Figure 1. Differences in endometrial and decidual NK cell populations. eNKs are comparatively cytotoxic, while dNKs are intentionally non-toxic and produce anti-inflammatory cytokines to preserve the maternal-fetal interface (Rao et. al, 2022).
Local Responses: T Cells
Different types of T cells are involved in immune responses, including Th1 and Th17 cells. Th1 cells cause the immune system to attack and phagocytose the antigen via IFNγ, while Th17 cells are responsible for inflammation via IL-17. These cells must be present in low quantities during pregnancy to avoid potential complications (Warning et. al, 2011).
Treg cells are a specific type of T cell that helps suppress the immune system’s activity to prevent fetal rejection. The mechanism used for this regulation is the suppression of CD4+ and CD8+ T cells (Sharma 2015). In fact, Tregs express a signature transcription factor, FoxP3, which activates the genes required for these suppressor functions. These cells also produce the cytokines IL-10 and TGF-β, which are both necessary immunosuppressive components to maintain the maternal-fetal interface. In the uterus, Tregs also mediate the inhibition of activated CD4+/CD25- T cell proliferation, and this requires cell-cell contact (Sharma 2015). Specifically, Treg cells constitutively express CTLA-4, which can interact with both antigen-presenting cells and activated T cells to inhibit the immunoactive response.
It follows that healthy pregnancies tend to have more Treg cells– meaning greater immune suppression around the fetus–and that these cells decrease pregnancy complications like preeclampsia.
Interestingly, trophoblasts—the cells that form the placenta and directly contact maternal tissues—influence cell differentiation and determine whether maternal T cells become Th1, Th2, Th17, or Treg cells. This occurs through the release of immunomodulatory cytokines, like IL-10 and TGF-β, and suggests that issues with trophoblast signaling disrupt the level of fetal tolerance (Warning et. al, 2011).
Conclusions
During pregnancy, both systemic responses and local changes in HLAs, NKs, and T cells accommodate and nourish the maternal-fetal interface. These responses coordinate to dampen immune responses and modulate the function of immune cells to act in an anti-inflammatory way. When these changes do not occur appropriately, immune and inflammatory responses can disrupt the materno-fetal graft and increase the likelihood of conditions like preeclampsia and spontaneous abortion.
Sources
Csabai, T., Pallinger, E., Kovacs, A., et al. (2020). Altered Immune Response and Implantation Failure in Progesterone-Induced Blocking Factor-Deficient Mice. Frontiers in Immunology (11). https://doi.org/10.3389/fimmu.2020.00349
Lee, J.Y., Lee, M., & Lee, S.K. (2011). Role of endometrial immune cells in implantation. Clinical and Experimental Reproductive Medicine 38(3). https://doi.org/10.5653/cerm.2011.38.3.119
Morley L.C. & Debant M., et al. (2021). Placental blood flow sensing and regulation in fetal growth restriction. Placenta (13). https://doi.org/10.1016/j.placenta.2021.01.007
Okada, H., Tsuzuki, T., & Murata, H. (2018). Decidualization of the human endometrium. Reproductive Medicine and Biology 17(3). https://doi.org/10.1002/rmb2.12088
Raghupathy, R., & Szekeres-Bartho, J. (2022). Progesterone: A Unique Hormone with Immunomodulatory Roles in Pregnancy. International Journal of Molecular Science(3), 1333. https://doi.org/10.3390/ijms23031333
Rao, A.V., Kurian, N.K., & Rao, K.A. (2022). Cytokines, NK cells and regulatory T cell functions in normal pregnancy and reproductive failures. American Journal of Reproductive Immunology(2). https://doi.org/10.1111/aji.13667
Sharma, S. (2015). Natural killer cells and regulatory T cells in early pregnancy loss. International Journal of Developmental Biology 58(0). https://doi.org/10.1387/ijdb.140109ss
Thelin, O. (2003). Pregnancy and the immune system: between tolerance and rejection. Science Direct(3), 179-184. https://www.sciencedirect.com/science/article/pii/S0300483X02006078
Warning, J.C., & McCracken, S.A. (2011). A balancing act: mechanisms by which the fetus avoids rejection by the maternal immune system. Reproduction(6), 715-724. https://doi.org/10.1530/REP-10-0360
Wei, X. & Yang, X. (2023). The central role of natural killer cells in preeclampsia. Frontiers in Immunology (14). https://doi.org/10.3389/fimmu.2023.1009867
Edited by Nandhini Parthasarathy
Read More

Kevin He '29
Apr 4, 2026
Universal Base-Edited CAR7 T-Cell Therapy to Treat Relapsed T-cell Acute Lymphoblastic Leukemia (T-ALL)
Base-edited CAR-T therapy offers new hope for relapsed T-ALL by inducing remission and bridging patients to stem cell transplant.

Keigo Fujita '29
Apr 3, 2026
Rejuvenating Senescent Dopaminergic Neurons: A Novel Therapeutic Approach to Parkinson's Disease
A novel therapeutic strategy for Parkinson's Disease may soon restore neuronal function without neuron elimination or replacement.

Nina Prakash
Mar 9, 2026
Nina Prakash's Senior Thesis Spotlight: Investigating the role of the serotonin 1B receptor in the neuroplastic effects of psilocybin in mice
Nina Prakash ’25 studies whether psilocybin’s neuroplastic effects depend on serotonin 1B rather than serotonin 2A.